 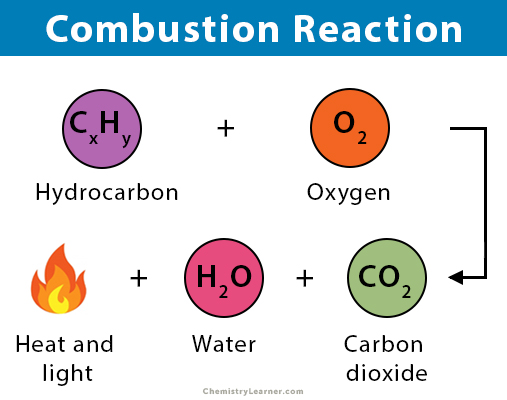
Example: Whenever you light propane (C3H8) in a gas barbecue, propane reacts with oxygen gas in a ratio of 1 to 5( of molecules) to produce carbon dioxide gas and water vapour in a ratio of 3 to 4. How many moles of O 2 are produced from the decomposition of 7.53 moles of H 2O 2?īecause both of the chemicals that are referenced in the question, O 2 and H 2O 2, are also present in the given reaction equation, a stoichiometric equality should be developed and applied when solving the problem. How to balance combustion reactions C H O text Calpha text Hbeta text Ogamma CHO denotes the generic chemical formula of the fuel. Introduction to Equations Definition A chemical equation is a symbolic representation of a chemical reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
